An understanding of cancer stem cells is critical in the treatment of cancer and the development of new cancer therapies. Cancer stem cells (CSCs) are a specific type of tumor cell that can have a major influence on the growth and development of the debilitating disease. As their name suggests, cancer stem cells possess the ability to differentiate and self-renew, with the potential to initiate, propagate, and spread cancer in patients and cause relapses after treatment.1 Therapeutics have to target and fight CSCs to effectively treat cancer. In addition to covering the basic biology underpinning CSCs, this piece will also give a brief overview of how innovative resources and services for the lab can facilitate targeting cancer stem cells in biomedical research and cancer therapeutics.
How do cancer stem cells develop?
Stem cells have the ability to freely differentiate into different types of mature tissues, such as muscles, bones, and organs – which are key to normal development patterns in the body.2 Stem cells continuously go through rounds of replication and maturation. While Stem cells have distinctive properties in order to prevent mutations, they can occur and with catastrophic consequences, as cancer is the likely result.2 CSCs arise due to mutations in the genomes of normal functioning stem cells. These cells are, thus, where the tumor originates and often make up a very small amount of the total tumor mass (around 0.01-2%) that can be difficult to target with therapeutics.2 Many existing cancer treatments focus efforts on the tumor mass as a whole and do not target these cells directly.
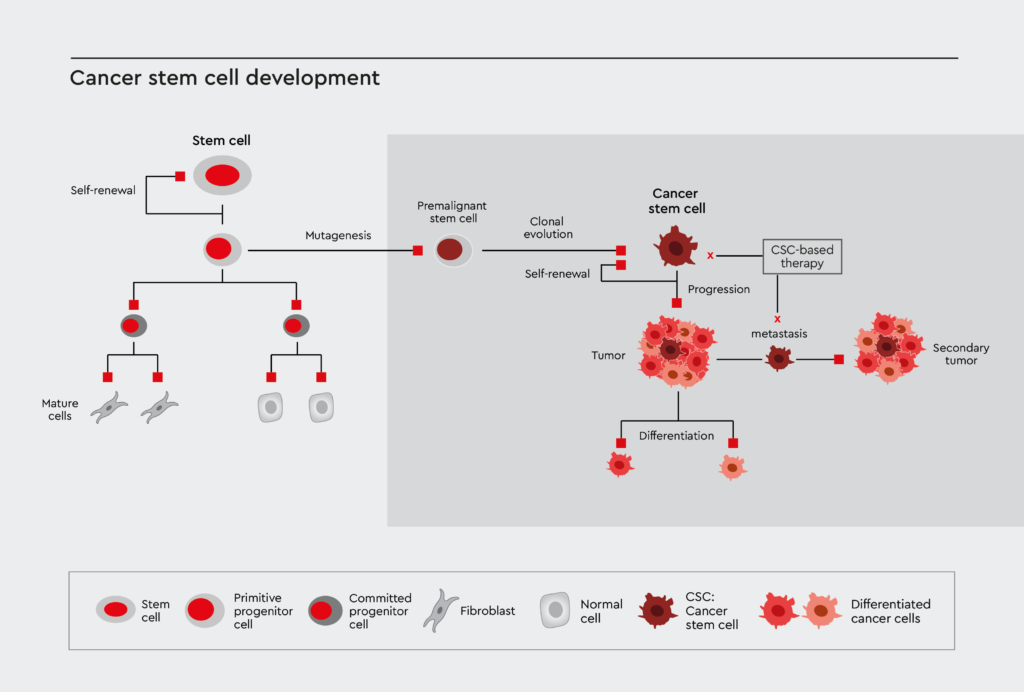
The role of cancer stem cells in tumor biology
Tumor initiation is driven by CSCs during the regeneration of tissues or in response to external carcinogens such as radiation or toxins that instigate malignant mutations.1 In cancer, mutations such as the overexpression of oncogenes and inactivation of tumor suppressors cause cells to undergo uncontrollable growth with debilitating consequences on normal cell growth and development.1 This uncontrolled propagation of cells forms a tumor. Specific properties of CSCs – in contrast to proliferative cancer cells – lead them to have a high potential to undergo unstable genetic changes. This means that tumors that are formed from CSCs can become more aggressive, invasive, and resistant to treatments as time goes on, eventually becoming metastatic and spreading to other tissues and areas in the body.1

Due to the critical role of CSCs in tumor initiation as well as disease recurrence, many researchers are focused on characterizing biomarkers for the cells so that treatment can be enhanced. While it is important to fight the tumor mass itself, remaining CSCs have the potential to develop metastases, and the cancer continues to spread. Thus, it is integral to find a way to use cancer stem cells as a target to improve and innovate personalized biomedical solutions to the deadly disease.
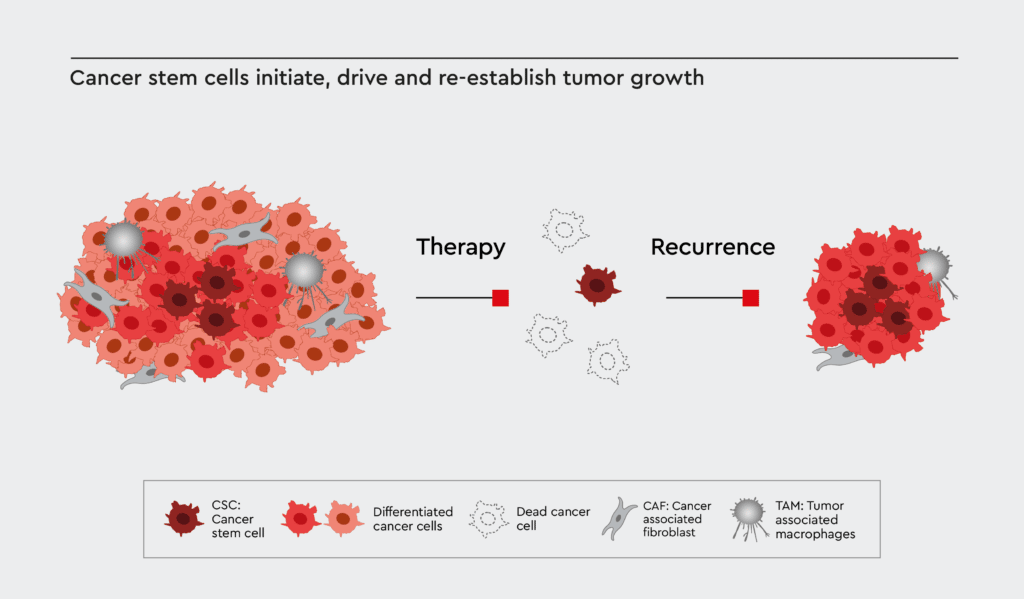
Current approaches to target cancer stem cells
Various approaches to target CSCs are being developed, but they still have their limitations. For example, disrupting signaling pathways relevant to these specific cells could be useful; however, CSCs share many pathways with regular stem cells, making this treatment option not specific.3 Targeting certain surface markers on these cells is another potential way to treat cancer. However, distinguishing specific biomarkers not expressed on normal stem cells is difficult and reduces treatment accuracy and efficacy.3 Treatment via drugs is also risky due to the different genetic and epigenetic landscape of each patient, as this could result in widely variable responses to the drug and severe side effects.4 Furthermore, using animal models is time-consuming, costly, and less clinically translatable to human systems.4
The primary issue for the successful development of CSC therapies is the ability to support the “stemness” of cancer stem cells outside of their in vivo physiological niche, which is central to developing suitable cell culture models for study. Traditional cell media used in tumor studies also support benign tumor cell growth and often display a loss in malignant cells needed for further study.4 Also, with standard approaches such as fluorescence-activated cell sorting (FACS) and magnetic-activated cell sorting (MACS), it is difficult and cumbersome to isolate and culture cancer stem cells in vitro. As CSCs are highly diverse and there is a lack of distinct and specific markers, the identification, isolation, and culture of the cells remain a challenge to scientists.5
Innovating solutions for working with cancer stem cells
As mentioned, it can be difficult to isolate and culture CSCs in vitro with standard approaches. Thus, working with CSCs requires innovative and specific solutions to address these laboratory workflow challenges. We are a leader in developing solutions for biomedical science. Our scientific experts provide researchers with resources to pursue answers and innovate treatments for pressing biological health challenges like cancer. Specifically, our Primary Cancer Culture System can be used for the isolation and culture of CSCs from tumor biopsies as well as to establish cancer cell lines.6
The Primary Cancer Culture System (PCCS) is an effective and reliable part of a toolbox of media, which consists of the PCCS, the 3D Tumorsphere Medium XF, and the Cancer Cell Line Medium XF, that selects CSCs and maintains them in culture for further study, specifically supporting the growth of CSCs. This selectivity preserves diverse cancerous cell populations while also removing the need for an extra cell sorting step in the laboratory workflow.4 The toolbox is composed of different functional media that selects CSCs from any starting material, establishes a primary cancer cell line, and keeps a defined and healthy CSC subpopulation in any kind of experimental setup such as 3D tumorspheres, co-cultivation with other non-tumor cells, screening-assays, long-term studies and alike.
For example, tumorspheres provide insight into tumor biology by serving as a biologically relevant model that is better reflective of the tumor and the dynamics of its microenvironment.5 Tumorspheres require CSCs, which drive tumor formation in culture much as they do in vivo, to be properly maintained outside of their physiological niche as well as be properly formed for a specific amount of time to ensure that they can be adequately studied – all of these characteristics are enabled by our PromoCell cancer media toolbox.5
Contact our experts
Enabled by the advancement of technology and a deeper understanding of the importance of CSCs, innovative life science solutions such as this cancer media toolbox can be developed to address the obstacles in targeting the cells. If you are a researcher or clinical scientist working in a laboratory with CSCs in oncology studies and therapeutics development, contact an expert here at PromoCell today to learn more about how to adapt your workflow to isolate and maintain CSCs in culture
References
- Walcher, L., et al. (2020). Cancer Stem Cells—Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Frontiers in Immunology, 11.
- Stem cells and cancer. (2022). Harvard.edu.
- Sun, H.-R., et al. (2019). Therapeutic Strategies Targeting Cancer Stem Cells and Their Microenvironment. Frontiers in Oncology, 9.
- Hof, C. (2017, December 11). Primary Cancer Cells: A Culture System Opens the Door to Individualized Therapy. PromoCell.
- Hof, C. (2018, April 17). A Home for Cancer Stem Cells: 3D Tumorspheres. PromoCell.
- Primary Cancer Culture System. (2022, August 10). PromoCell.






