인류의 건강과 생명공학 발전을 위해 봉사하는 기업 드림셀
We Serving the Health and Biotechnology of Humanity
We Serving the Health and Biotechnology of Humanity
제품코드 : G9951
제품정보 : Inflammation Assay
The Caspase-Glo® 1 Inflammasome Assay is a simple, homogeneous, bioluminescent method to selectively measure the activity of caspase-1, an essential component of the inflammasome.
Inflammasomes are protein complexes induced by diverse inflammatory stimuli. Innate immune cells respond to pathogens and other danger signals with inflammasome formation and conversion of procaspase-1 zymogen into catalytically active caspase-1. Caspase-1 activation results in the processing and release of cytokines IL-1β and IL-18, as well as pyroptosis, an immunogenic form of cell death. The Caspase-Glo® 1 Assay is sensitive enough to measure caspase-1 activity directly in cells or in medium in multiwell plates.
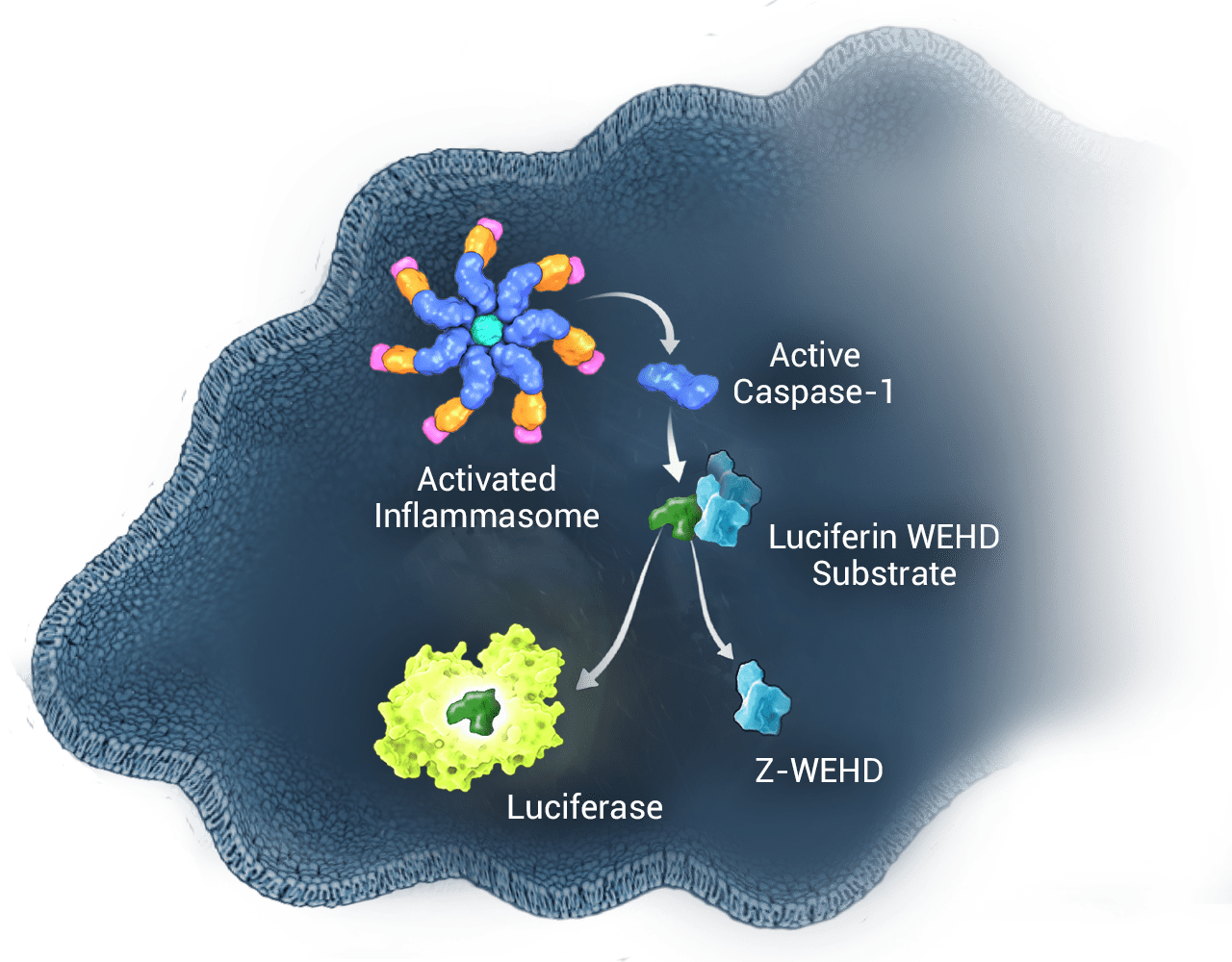
Following caspase cleavage of the Z-WEHD substrate (Z-WEHD-aminoluciferin), a substrate for luciferase (aminoluciferin) is released, resulting in luciferase activity and generation of light by a proprietary, thermostable, recombinant luciferase.

No sample preparation or manipulation required. Simply add, mix and then measure luminescence after only one hour. You can get results faster in fewer steps compared to conventional Western blot and ELISA analyses. The stable luminescent signal and large volume (100ml) format supports high-throughput screening applications.

The selective caspase-1 substrate (Z-WEHD) enables detection of catalytically active caspase-1 in cells or culture media and quantitative measurement of inflammasome activity. Inclusion of the proteasome inhibitor MG-132 in the reagent eliminates non-specific proteasome substrate cleavage, enabling sensitive detection of caspase-1. The assay includes a caspase-1 specific inhibitor (Ac-YVAD-CHO) to confirm specific activity in parallel samples and distinguish caspase-1 activity from other caspases.
Inflammation and apoptosis caspases at equimolar concentrations were tested with the assay, +/– Ac-YVAD-CHO, and caspase activity was measured after 20 minutes. Ac-YVAD-CHO at 1µM inhibited 99% of the caspase-1 activity, whereas it had minimal effect on the other cross-reacting caspases (5, 3 and 6).

NLRP3 plays a key role in immune sensing by initiating the assembly of an inflammasome in response to various danger signals. The NLRP3 inflammasome triggers caspase-1 activation and IL-1β cytokine secretion, eventually resulting in an inflammatory, pyroptotic cell death. The Caspase-Glo® 1 Assay allows you to easily measure the effect of NLRP3 inhibitors on caspase-1 activation in primary cells.

THP-1 cells were differentiated for 3 days with PMA. Medium was replaced and MCC950 (NLRP3 inflammasome inhibitor), was added followed by LPS or vehicle. Half of the culture medium (50µl) was removed after 3 hours of LPS and tested with Caspase-Glo® 1 Reagent or Caspase-Glo® 1 Reagent + YVAD-CHO.

PBMCs were plated, then treated with MCC950 followed by LPS or vehicle overnight. Following the overnight treatment, cells were treated with nigericin or vehicle. Nigericin causes potassium efflux and significant caspase-1 activation after LPS priming. Half of the culture medium (50µl) was then removed and tested with Caspase-Glo® 1 Reagent or Caspase-Glo® 1 Reagent + YVAD-CHO. Luminescence was read for both after 1 hour. Caspase-Glo® 1 signal demonstrates a dose-dependent inhibition of caspase-1 activity by the NLRP3 inflammasome inhibitor.
Caspase-Glo® 1 Inflammasome Assay monitors caspase-1 activity in peripheral blood mononuclear cell (PBMC) supernatant. Cells were stimulated with or without LPS then subsequently stimulated with ATP for 30 minutes. Cells were then pelleted and supernatant was assayed for caspase-1 activity.

The Caspase-Glo® 1 Assay is sensitive enough to detect caspase-1 activity from a small volume of transferred medium. This leaves cells intact for other assays, such as cell viability or cytotoxicity, so you can get more data per sample.



Caspase-Glo® 1 Inflammasome Assay Multiplexed with Cell Viability and Cell Death Assays. THP-1 cells were grown in RPMI 1640 medium supplemented with 10% FBS in a 37°C incubator with 5% CO2. Cells were added to plates at 5 x 105 cells/ml in 100µl of medium and differentiated with PMA (20nM, 3 days) in 96-well plates followed by treatment with flagellin (1µg/ml, 1 hour) or nigericin (20µM, 2 hours). In Panel A CellTox™ Green Reagent, Caspase-Glo® 1 Reagent or Caspase-Glo® 1 YVAD-CHO Reagent was added to the cells, and fluorescence (CellTox™ Green) or